SSINA: Stainless Steel: Corrosion. Definition: The combination of tensile stress and a specific corrosive environment can crack stainless steels. This mode of attack is termed stress corrosion cracking (SCC). The most common environmental exposure condition responsible for SCC of stainless steels is the presence of chlorides. Although no stainless steel grade is totally immune to chloride SCC, the relative resistance of stainless steels varies substantially.

Influence of Alloy Composition: The relative resistance to chloride SCC is dependant on the stainless steel family. The austenitic family of stainless steels is the most susceptible. The resistance of austenitic stainless steels to SCC is related to the nickel content of the steel. The most susceptible austenitic grades have nickel contents in the range of 8 to 1. Therefore, standard grades such as 3. L and 3. 16/3. 16.
L are very susceptible to this mode of attack. Austenitic grades with relatively high nickel and molybdenum contents such as alloy 2. L, and the 6% molybdenum super austenitic grades have substantially better chloride SCC resistance. The ferritic family of stainless steels, which includes grades such as type 4.



STAINLESS STEEL 408. Stainless steela ahow good resistance to all concentrations of ammonia. STRESS CORROSION CRACKING. CORROSION: Chloride Stress Corrosion Cracking. Although no stainless steel grade is totally immune to chloride SCC. use and avoidance of stress corrosion cracking, and Stainless Steel in Swimming Pool Buildings.. Stress Corrosion Cracking. system.Chloride stress corrosion cracking in austenitic stainless steel is characterized by the multi. and occurs in austenitic stainless steel under tensile stress in the.
SCC. The duplex stainless steel with their dual austenite/ferrite microstructures has a resistance that is in between that of the austenite and ferrite grades. Corrosion Testing. The relative resistance of a stainless steel to chloride SCC is often quantified by the use of standard boiling salt solutions. The following table summarizes the results of testing in boiling salt solutions of 2. Na. Cl (sodium chloride), 3. Li. Cl (lithium chloride), and 4. Mg. Cl. 2 (magnesium chloride). The boiling Li. Cl and Mg. Cl. 2 test solutions are very aggressive relative to practical applications and only austenitic alloys with compositions that approach those of nickel- base alloys will routinely resist cracking in these test solutions.
Table 1: Relative chloride SCC resistance measured using fully immersed U- bend specimens in standard boiling salt solutions. Taken from producer data)Alloy. Mg. Cl. 23. 3% Li.
Cl. 26% Na. Cl. Austenitic SSTType 3. L (S3. 04. 03)SCCSCCSCCType 3. L (S3. 16. 03)SCCSCCSCC9.
L (N0. 89. 04)SCCSCCNo Cracking. Mo SSTSCCSCCNo Cracking. Alloy 2. 0 (N0. 80. SCCNo Cracking. No Cracking.
Austenitic stainless steel UNS S31254 are reported in this paper. Experimental. ring specimens to obtain max. stress. chloride stress corrosion cracking. Stress Corrosion Cracking of Stainless Steels in Chlorides Environments. concentration and oxygen level for the various stainless steel families. EXECUTIVE SUMMARY Objectives Chloride stress corrosion cracking (CLSCC) is one of the most common reasons why austenitic stainless steel pipework and vessels deteriorate in the chemical processing and petrochemical industries.
Duplex SST2. 20. S3. 22. 05)SCCNo Cracking. No Cracking. 25. S3. 25. 50)SCC- -- No Cracking. SCC- -- No Cracking. Ferritic SST4. 39 (S4. No Cracking. No Cracking.
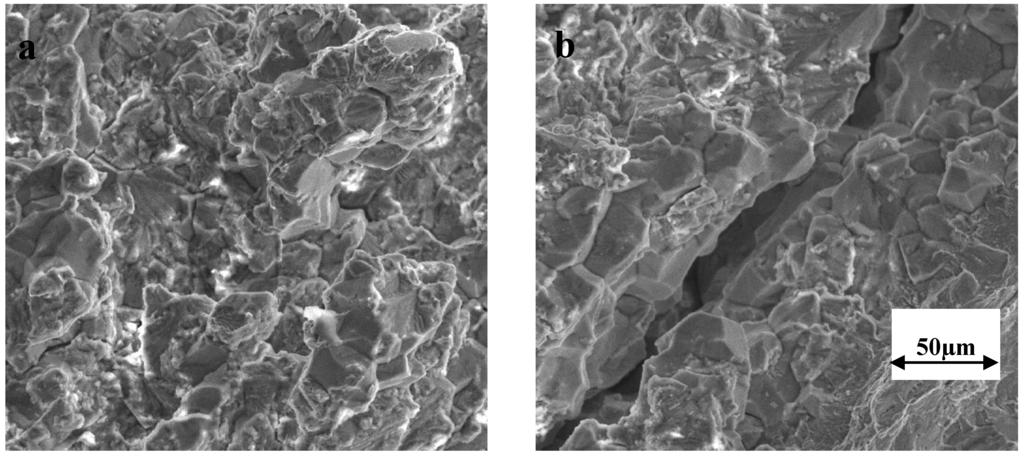
No Cracking. 44. S4. 44. 00)No Cracking. No Cracking. No Cracking. Crack Appearance. The typical crack morphology for chloride stress corrosion cracking consists of branched transgranular cracks. Figure 1 shows the cracking that occurred on a 6.
Mo super austenitic stainless steel (N0. F (2. 60 °C) Figure 1: Typical appearance of chloride stress corrosion cracking. Photo courtesy of TMR Stainless. Environmental Factors: The environmental factors that increase the cracking susceptibility include higher temperatures, increased chloride content, lower p. H, and higher levels of tensile stress. Temperature is an important variable. When stainless steels are fully immersed, it is rare to see chloride stress corrosion cracking at temperatures below 6. C (1. 50 °F). There is a synergistic relationship between dissolved oxygen and the chloride level. If the oxygen level is reduced to the 0. L and 3. 16. L. The normal solubility of O2 in water at room to moderate temperatures (e.
F/6. 0°C) is 4. 5 to 8 ppm at atmospheric pressure. In actual service environments, evaporation can produce local build- up of aggressive corrosive substances, such as chlorides and the H+ ions, resulting in conditions that are substantially more aggressive. Under severe evaporative conditions, stainless steels can crack at temperatures well below the thresholds measured under conditions where there is full immersion. Because of this, one must use caution when specifying materials for applications that involve the evaporation of chloride- bearing solutions on hot stainless steel surfaces. The Materials Technology Institute (MTI) of the Chemical Process Industry has reviewed literature and collected case histories to define guidelines for the chloride SCC susceptibility of types 3.
L and 3. 16. L stainless steel in neutral water environments. Figure 2 shows the cracking threshold for 3. L and 3. 16. L stainless steel as a function of temperature and chloride content. The level of chlorides required to produce cracking is relatively low. Failures have been reported in environments with as little as 1. This is particularly true for environments having concentrating (evaporating) mechanisms such as wet/dry interfaces or a film of solution in immediate contact with a heat- rejecting surface. In these situations, a few ppm of chlorides in the bulk solution can concentrate to hundreds of ppm in the area of evaporation. Figure 2: Cracking threshold for 3. The cracking threshold of a 6.
Mo super austenitic stainless steel (UNS N0. Figure 3. The temperature thresholds are well above the 2. F (1. 00°C) range, indicating that exposures to atmospheric boiling in neutral chloride solutions are very unlikely to produce cracking.
Figure 3: Cracking threshold for a 6. Mo super austenitic steel ( UNS N0.
Na. Cl solutions. Courtesy of TMR Stainless. Swimming Pools. As was noted above, it is rare to see chloride stress corrosion cracking at temperatures below 6. C (1. 50 °F). Elevated load bearing applications in interior swimming pools are an exception to this rule and have a unique set of conditions. For more information, please read, Successful Stainless Swimming Pool Design, Stainless steels for swimming pool building applications – selection, use and avoidance of stress corrosion cracking, and Stainless Steel in Swimming Pool Buildings.
Additional References. There are numerous NACE papers and stainless steel producer brochures on this topic. Additionally, the following industry association brochures are suggested as general references.
Nickel Institute brochure No. High Performance Stainless Steels.
Nickel Institute brochure 1. Practical Guidelines for the Fabrication of High Performance Austenitic Stainless Steels. International Molybdenum Association brochure Practical Guidelines for the Fabrication of Duplex Stainless Steelsh.
Stainless Steel - Corrosion Resistance. Written by AZo. MJan 8 2. Topics Covered. Introduction. General Corrosion.
Pitting Corrosion. Pitting Resistance Equivalent number (PRE)Crevice Corrosion. Stress Corrosion Cracking (SCC)Sulfide Stress Corrosion Cracking (SSC) Stress Level. Environment. Temperature. Intergranular Corrosion. Galvanic Corrosion. Contact Corrosion.
Passivation and Pickling. Introduction. Corrosion resistance is one of the main advantages of the application of stainless steels. However, in some cases, stainless steels suffer certain types of corrosion and therefore precautions should be taken in choosing a suitable steel grade for any application. Based on the applications, corrosion results in a number of problems as follows.
Pitting or corrosion products can deteriorate the appearance of a product by affecting the decorative surface finish. Corrosion leads to the formation of rust or scale, which contaminates the material. This usually occurs in food processing systems. Corrosion reduces the cross section of structural members, causing loss of strength of the structure.
Perforation of pipes or tanks, leading to leakage of gases or fluids. Stainless steels are subjected to one of the following corrosion phenomena. Contact corrosion. Galvanic corrosion. Intergranular corrosion.
Stress corrosion cracking. Sulfide stress corrosion cracking. Crevice corrosion. Pitting corrosion.
General corrosion. General Corrosion. Corrosion is a process of uniform removal of material through dissolution - e. In this instance, design and life span of the material is based on published data. Description on the gradual removal of metal can be obtained from the published data list. Further, the list contains tables of corrosion resistance to various chemicals provided by several organizations, a large collection of charts, and technical papers published by stainless steel suppliers and manufacturers. Pitting Corrosion.
In some cases - which involve acidic conditions, moderately high temperatures and high chloride concentration - localized corrosion takes place causing perforation of fittings and pipes. This type of corrosion can penetrate the cross section of the material. Steel grades containing high amounts of chromium, nitrogen and molybdenum have high resistance to pitting corrosion.
Pitting Resistance Equivalent number (PRE). The pitting resistance equivalent number (PRE) indicates the rate of the pitting resistance of stainless steels.
The PRE can be calculated using the following equation: PRE = %Cr + 3. Mo + 1. 6 x %N. Pitting corrosion needs to be taken seriously due to the fact that when a pit is formed on a surface of the steel, it will continue to develop in the surrounding area. The tendency for a particular steel to be attacked by pitting corrosion can be evaluated in the laboratory. A number of standard tests have been devised - the most common of which is that given in ASTM G4. A graph can be drawn giving the temperature at which pitting corrosion is likely to occur, as shown in Figure 1. The extent to which a particular steel can be affected by pitting corrosion can be determined in the laboratory. Although a number of standard test procedures have been devised, the most common procedure is given in ASTM G4.
Figure 1 shows a graph providing the possible temperatures at which pitting corrosion can occur. Figure 1 - illustrates the temperature at which pitting corrosion is likely to occur.
This graph is based on a conventional ferric chloride laboratory test. Crevice Corrosion. A protective oxide layer present on the surface of stainless steel determines its corrosion resistance. However this oxide layer is vulnerable to damage under certain conditions, such as in reducing acids, or in certain combustion mechanism having a reduced atmosphere. Certain areas of the component, such as sharp re- entrant corners or overlapping surfaces, have the tendency to form crevices that promote corrosion. Crevices need to be of sufficient width to allow entry of the corrodent, and at the same time narrow to ensure that corrodent remains in its place. Accordingly crevice corrosion generally occurs in gaps of a few micrometers width, and it does not occur in groves that allow circulation of the corrodent.
This problem can be resolved by avoiding the formation of crevices, or by keeping them open. Crevice corrosion is similar to pitting corrosion, and alloys resistant to one type of corrosion will be resistant to the other. Crevice corrosion occurs at lower temperatures when compared to pitting corrosion. Stress Corrosion Cracking (SCC).
Stainless steels can be subjected to rapid and very serious corrosion under the conditions of stress and corrosive environments. The stresses can be tensile, resulting from the application of loads, or residual stresses from the method of fabrication. The most damaging environment is the chloride environment with high temperatures. Austenitic group of steels is more commonly affected by corrosion due to chlorides.
The duplex stainless steels, on the other hand, are more resistant to stress corrosion cracking (SCC) than austenitic grades. The ferritic grades are free of SCC. It has been found that SCC resistance can be improved in some cases with the application of compressive stress to the material at risk. This can be performed by shot peening the material surface, or by annealing the surface to relieve tensile stresses. Under certain conditions chloride stress corrosion failures occur at lower temperatures of 3. C. Such failures take place in the warm, moist environment above indoor chlorinated swimming pools, where stainless steel fixtures are often used to suspend items such as ventilation ducting. Failures due to stress corrosion at higher temperatures with chloride levels as low as 1.
Sulfide Stress Corrosion Cracking (SSC). Material resistance to sulfide stress corrosion cracking is of prime importance for oil and gas industrial applications. Sulfide stress corrosion cracking occurs due to conjoint action of chloride and hydrogen sulfide, presence of tensile stresses and non- linear relationship with temperature.
The three main factors of sulfide stress corrosion cracking are stress level, environment and temperature. Stress Level. In some cases a threshold stress can be observed in each material. Some reports show a continuous reduction threshold stress with increasing concentration of H2. S. According to NACE specification MR0. HRC to avoid sulfide stress corrosion cracking.
Environment. Hydrogen sulfide, chloride and p. H are the principal agents of sulfide stress corrosion cracking. Temperature. The concentration of chloride increases with increasing temperatures, while the effect of hydrogen decreases - owing to its increased mobility in the ferrite matrix. As a result, maximum susceptibility can be observed in the range of 6. C. Heat tint at welds, presence of cold work, surface condition and amount of ferrite are some of the observed secondary factors that promote sulfide stress corrosion cracking. Intergranular Corrosion. Intergranular corrosion is a process of rapid and localized corrosion, resulting from a defective microstructure such as carbide precipitation.
Upon exposing austenitic steels to temperatures of 4. C for a time period, or heating the steel to high temperatures followed by slow cooling, chromium carbide particles are formed along the grain boundaries of the steel. This deteriorates the surrounding environment of the metal surface, thereby reducing the corrosion resistance of the steel. Steel is termed as “sensitized” in this condition. Carbide precipitation generally depends on the time, temperature and carbon content. The most critical temperature range is about 7. C, at which 0. 0. Steels subjected to carbide precipitation can be reclaimed by heating them above 1.
C followed by water quenching, to retain chromium and carbon in solution thereby preventing the formation of carbides. However, this treatment is not suitable for welded or heated structures, and hence special steel grades have to be designed to avoid this problem. Such special grades are grade 3. Use of extra low carbon grades, such as grades 3. L and 3. 16. L, is another method to overcome intergranular corrosion.
These grades have extremely low carbon levels and hence they have more resistance to carbide precipitation. Sensitized austenitic stainless steels are free of intergranular corrosion in most of the environments, such as alkaline salt solution, glacial acetic acid at room temperature, and fresh water. It should also be noted that high temperature strength of stainless steels remains unaffected by grain boundary carbides. Grades used for high temperature applications often have high carbon contents, which in turn enhances their creep resistance and high temperature strength. Galvanic Corrosion. Galvanic corrosion is an electrochemical process, which involves flow of electric current.
It occurs due to the galvanic effect caused by the contact of two dissimilar metals in an electrically conductive liquid. The following are the three major conditions for galvanic corrosion. Surfaces of the metal should be bridged by an electrolyte. Two dissimilar metals must be in electrical contact with each other.
Two metals should be separated from each other on the galvanic series. Figure 2 - illustrates galvanic series of metals in flowing seawater. Avoiding mixed metal fabrications can prevent galvanic corrosion. However, this is not practically possible and, hence, cutting the electrical contact between metals is usually recommended for the prevention of galvanic corrosion. Electrical contact can be cut by using rubber or plastic washers or sleeves, or by using protective hoods or draining the electrolyte.
Galvanic corrosion effect is also based on the areas of dissimilar metals. The corrosive effect may be negligible if the area of less noble material is larger than that of more noble material.
 RSS Feed
RSS Feed
